The Mechanochemical Coupling Mechanism of Matrix Stiffnesses and Growth Factors Driving the Epithelial-Mesenchymal Transition
-
摘要:
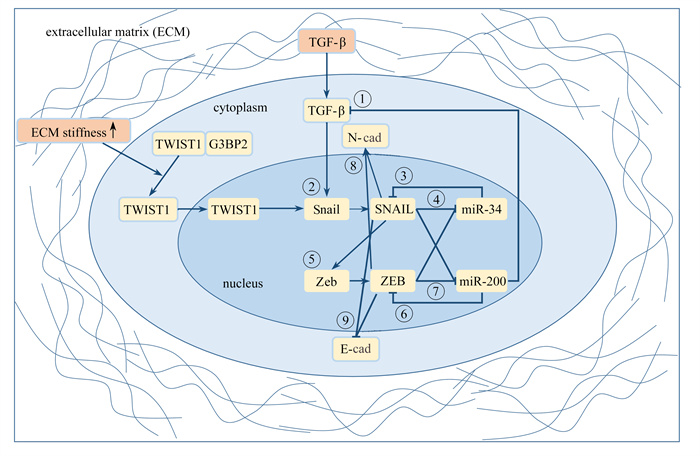
上皮-间质转化(epithelial to mesenchymal transition, EMT)是胚胎发育、伤口愈合、癌症发展等生理、病理过程中的关键步骤,使细胞从紧密黏附在一起的上皮状态转变为分散排布的间质状态. 该文提出了一个基质刚度和生长因子协同驱动EMT的核心调控回路模型,发现在EMT过程中,基质刚度和生长因子通过协同调控EMT激活转录因子(EMT-activating transcript factors, EMT-TFs)来改变细胞间力学黏附分子E/N-钙黏素的表达,从而影响EMT的进程与可逆性. 该模型揭示了力学和化学因素的协同作用对EMT过程中细胞间力学黏附的影响机制,为研究癌症等疾病的发生、发展机制和防治策略奠定了理论基础.
Abstract:The epithelial-mesenchymal transition (EMT) is a critical step in physiological and pathological processes such as the embryonic development, the wound healing, and the cancer progression, wherein cells transition from a tightly adherent epithelial state to a dispersed mesenchymal state. An EMT core circuit model driven by the synergistic regulation of matrix stiffnesses and growth factors was proposed. The results show that, during the EMT, the matrix stiffnesses and growth factors collaboratively regulate the expression of the E/N-cadherin, a typical cell-cell adhesion molecule, by modulating the EMT-activating transcription factors, thus influencing the progression and reversibility of the EMT. The model elucidates the mechanism of synergistic interactions between mechanical and chemical factors on cell-cell adhesion during the EMT, laying a theoretical foundation for understanding the occurrence, development mechanisms, and preventive strategies against diseases such as cancer.
-
Key words:
- mechanical modeling /
- mechanochemical coupling /
- epithelial-mesenchymal transition /
- cell-cell adhesion
edited-byedited-by1) (我刊编委林敏来稿) -
图 4 在不同外源性TGF-β相对浓度(ρTGF, ext0)诱导细胞EMT发生10天后,将细胞置于相应外源性TGF-β相对浓度(ρTGF, ext1)下培养10天,细胞中钙黏素相对浓度的变化
Figure 4. Relative expressions of cadherins in cells firstly treated with various exogenous TGF-β levels (ρTGF, ext0) for 10 days and then with indicated exogenous TGF-β levels (ρTGF, ext1) for another 10 days
图 8 上皮细胞在一定外源性TGF-β相对浓度(ρTGF, ext0)培养10天后,再撤去外源性TGF-β培养10天,N-钙黏素相对浓度的最大值和最终值随不同外源性TGF-β相对浓度和基质刚度的变化相图
Figure 8. Phase diagrams showing N-cadherin maxima and end-point values at different matrix stiffnesses and exogenous TGF-β levels (ρTGF, ext0). The simulations correpond to epithetial cells firstly treated with various exogenous TGF-β levels for 10 days and then without exogenous TGF-β for another 10 days
表 1 完全EMT后细胞中相关分子的相对浓度
Table 1. Relative expressions of related molecules in the full EMT state
notation meaning value reference ρTGF, M relative expression of endogenous TGF-β in mesenchymal cells 10 [19] ρRsnail, M relative expression of Snail mRNA in mesenchymal cells 5 [20, 43] ρSNAIL, M relative expression of SNAIL in mesenchymal cells 10 [20, 43] ρmiR34, M relative expression of miR-34 in mesenchymal cells 0.15 [20] ρRzeb, M relative expression of Zeb mRNA in mesenchymal cells 5 [20] ρZEB, M relative expression of ZEB in mesenchymal cells 10 [20] ρmiR200, M relative expression of miR-200 in mesenchymal cells 0.15 [20] ρEcad, M relative expression of E-cadherin in mesenchymal cells 0.13 [20, 43] ρNcad, M relative expression of N-cadherin in mesenchymal cells 8 [20, 43] 表 2 模型参数
Table 2. Parameters used in the model
notation meaning value kgT/h-1 production rate of TGF-β 0.25 JT Michaelis constant of miR-200-inhibited production of TGF-β 0.29 kdT/h-1 degradation rate ofTGF-β 0.02 kg0s/h-1 basic production rate of Snail mRNA 0.26 kgs/h-1 TGF-β-dependent production rate of Snail mRNA 1.04 Js Michaelis constant of TGF-β-dependent Snail mRNA production 5 kds/h-1 degradation rate of Snail mRNA 0.26 kgS/h-1 production rate of SNAIL 0.16 JS Michaelis constant of SNAIL production 1 kdS/h-1 degradation rate of SNAIL 0.08 kg34/h-1 production rate of miR-34 0.53 J34S Michaelis constant of SNAIL-dependent miR-34 production 10 J34Z Michaelis constant of ZEB-dependent miR-34 production 4.5 kd34/h-1 degradation rate of miR-34 0.5 kgz/h-1 production rate of Zeb mRNA 1.04 Jz Michaelis constant of Zeb mRNA production 2 kdz/h-1 degradation rate of Zeb mRNA 0.2 kgZ/h-1 production rate ZEB 0.2 JZ Michaelis constant of ZEB production 1 kdZ/h-1 degradation rate of ZEB 0.1 kg200/h-1 production rate of miR-200 0.53 J200S Michaelis constant of SNAIL-dependent miR-200 production 4.5 J200Z Michaelis constant of ZEB-dependent miR-200 production 10 kd200/h-1 degradation rate of miR-200 0.5 kg1E/h-1 SNAIL-dependent production rate of E-cadherin 0.011 JES Michaelis constant of SNAIL-dependent E-cadherin production 3.7 kg2E/h-1 ZEB-dependent production rate of E-cadherin 0.011 JEZ Michaelis constant of ZEB-dependent E-cadherin production 3.7 kdE/h-1 degradation rate of E-cadherin 0.02 kg1N/h-1 SNAIL-dependent production rate of N-cadherin 0.086 JNS Michaelis constant of SNAIL-dependent N-cadherin production 2.76 kg2N/h-1 ZEB-dependent production rate of N-cadherin 0.086 JNZ Michaelis constant of ZEB-dependent N-cadherin production 2.76 kdN/h-1 degradation rate of N-cadherin 0.02 $\tilde{k}_{\mathrm{CN}}$ relative import rate of TWIST1 into nucleus 10 $\tilde{k}_{\mathrm{g}}^{\mathrm{TG}}$ relative binding rate of TWIST1 to G3BP2 100 E0/kPa stiffness where TWIST1 phosphorylation reaches the half maximum level 0.1 -
[1] NIETO M A, HUANG R Y J, JACKSON R A, et al. EMT: 2016[J]. Cell, 2016, 166(1): 21-45. doi: 10.1016/j.cell.2016.06.028 [2] 孙玉川, 李红, 罗庆, 等. 肿瘤组织力学异质性与肿瘤细胞的上皮-间质转化[J]. 医用生物力学, 2021, 36(4): 658-663. https://www.cnki.com.cn/Article/CJFDTOTAL-YISX202104027.htmSUN Yuchuan, LI Hong, LUO Qing, et al. Mechanical heterogeneity of tumor tissues and epithelial-mesenchymal transition of tumor cells[J]. Journal of Medical Biomechanics, 2021, 36(4): 658-663. (in Chinese) https://www.cnki.com.cn/Article/CJFDTOTAL-YISX202104027.htm [3] 刘扬, 王金佩, 黄国友, 等. 肿瘤上皮-间质转化(EMT)的生物力学特性研究进展[J]. 西南民族大学学报(自然科学版), 2020, 46(6): 571-577. https://www.cnki.com.cn/Article/CJFDTOTAL-XNMZ202006005.htmLIU Yang, WANG Jinpei, HUANG Guoyou, et al. Research advances in biomechanical properties of EMT in tumor cells[J]. Journal of Southwest University for Nationalities (Natural Science Edition), 2020, 46(6): 571-577. (in Chinese) https://www.cnki.com.cn/Article/CJFDTOTAL-XNMZ202006005.htm [4] FONT-NOGUERA M, MONTEMURRO M, BENASSAYAG C, et al. Getting started for migration: a focus on EMT cellular dynamics and mechanics in developmental models[J]. Cells & Development, 2021, 168: 203717. [5] THIERY J P, ACLOQUE H, HUANG R Y J, et al. Epithelial-mesenchymal transitions in development and disease[J]. Cell, 2009, 139(5): 871-890. doi: 10.1016/j.cell.2009.11.007 [6] STEMMLER M P, ECCLES R L, BRABLETZ S, et al. Non-redundant functions of EMT transcription factors[J]. Nature Cell Biology, 2019, 21(1): 102-112. doi: 10.1038/s41556-018-0196-y [7] PUISIEUX A, BRABLETZ T, CARAMEL J. Oncogenic roles of EMT-inducing transcription factors[J]. Nature Cell Biology, 2014, 16(6): 488-494. doi: 10.1038/ncb2976 [8] HAO Y, BAKER D, TEN D P. TGF-β-mediated epithelial-mesenchymal transition and cancer metastasis[J]. International Journal of Molecular Sciences, 2019, 20(11): 2767. doi: 10.3390/ijms20112767 [9] BOARETO M, JOLLY M K, GOLDMAN A, et al. Notch-jagged signalling can give rise to clusters of cells exhibiting a hybrid epithelial/mesenchymal phenotype[J]. Journal of the Royal Society Interface, 2016, 13(118): 20151106. doi: 10.1098/rsif.2015.1106 [10] 刘祉宁, 桑晨. 炎性因子引起器官纤维化及上皮-间充质转化机制的研究进展[J]. 生命科学, 2018, 30(8): 868-875. https://www.cnki.com.cn/Article/CJFDTOTAL-SMKX201808010.htmLIU Zhining, SANG Chen. Roles of proinflammatory cytokines in organ fibrosis and epithelial-mesenchymal transition[J]. Chinese Bulletin of Life Sciences, 2018, 30(8): 868-875. (in Chinese) https://www.cnki.com.cn/Article/CJFDTOTAL-SMKX201808010.htm [11] DESPRAT N, SUPATTO W, POUILLE P A, et al. Tissue deformation modulates twist expression to determine anterior midgut differentiation in drosophila embryos[J]. Developmental Cell, 2008, 15(3): 470-477. doi: 10.1016/j.devcel.2008.07.009 [12] 张众, XIAO G G S, 石宇, 等. 细胞生命进程中microRNA调控的意义[J]. 临床与实验病理学杂志, 2012, 28(5): 477-481. doi: 10.3969/j.issn.1001-7399.2012.05.001ZHANG Zhong, XIAO G G S, SHI Yu, et al. Significance of microRNA regulation in cell life process[J]. Chinese Journal of Clinical and Experimental Pathology, 2012, 28(5): 477-481. (in Chinese) doi: 10.3969/j.issn.1001-7399.2012.05.001 [13] FENG X, WANG Z, FILLMORE R, et al. MiR-200, a new star miRNA in human cancer[J]. Cancer Letters, 2014, 344(2): 166-173. doi: 10.1016/j.canlet.2013.11.004 [14] WELLNER U, SCHUBERT J, BURK U C, et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs[J]. Nature Cell Biology, 2009, 11(12): 1487-1495. doi: 10.1038/ncb1998 [15] CHEN L, GIBBONS D L, GOSWAMI S, et al. Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression[J]. Nature Communications, 2014, 5(1): 5241. doi: 10.1038/ncomms6241 [16] SIEMENS H, JACKSTADT R, HVNTEN S, et al. miR-34 and SNAIL form a double-negative feedback loop to regulate epithelial-mesenchymal transitions[J]. Cell Cycle, 2011, 10(24): 4256-4271. doi: 10.4161/cc.10.24.18552 [17] HAHN S, JACKSTADT R, SIEMENS H, et al. SNAIL and miR-34a feed-forward regulation of ZNF281/ZBP99 promotes epithelial-mesenchymal transition[J]. The EMBO Journal, 2013, 32(23): 3079-3095. doi: 10.1038/emboj.2013.236 [18] GREGORY P A, BRACKEN C P, SMITH E, et al. An autocrine TGF-β/ZEB/miR-200 signaling network regulates establishment and maintenance of epithelial-mesenchymal transition[J]. Molecular Biology of the Cell, 2011, 22(10): 1686-1698. doi: 10.1091/mbc.e11-02-0103 [19] TIAN X J, ZHANG H, XING J. Coupled reversible and irreversible bistable switches underlying TGFβ-induced epithelial to mesenchymal transition[J]. Biophysical Journal, 2013, 105(4): 1079-1089. doi: 10.1016/j.bpj.2013.07.011 [20] ZHANG J, TIAN X J, ZHANG H, et al. TGF-β-induced epithelial-to-mesenchymal transition proceeds through stepwise activation of multiple feedback loops[J]. Science Signaling, 2014, 7(345): ra91. [21] WEI S C, FATTET L, TSAI J H, et al. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through a TWIST1-G3BP2 mechanotransduction pathway[J]. Nature Cell Biology, 2015, 17(5): 678-688. doi: 10.1038/ncb3157 [22] LEIGHT J L, WOZNIAK M A, CHEN S, et al. Matrix rigidity regulates a switch between TGF-β1-induced apoptosis and epithelial-mesenchymal transition[J]. Molecular Biology of the Cell, 2012, 23(5): 781-791. doi: 10.1091/mbc.e11-06-0537 [23] LU M, JOLLY M K, LEVINE H, et al. MicroRNA-based regulation of epithelial-hybrid-mesenchymal fate determination[J]. Proceedings of the National Academy of Sciences, 2013, 110(45): 18144. doi: 10.1073/pnas.1318192110 [24] TRIPATHI S, CHAKRABORTY P, LEVINE H, et al. A mechanism for epithelial-mesenchymal heterogeneity in a population of cancer cells[J]. PLOS Computational Biology, 2020, 16(2): e1007619. doi: 10.1371/journal.pcbi.1007619 [25] BOCCI F, GEARHART-SERNA L, BOARETO M, et al. Toward understanding cancer stem cell heterogeneity in the tumor microenvironment[J]. Proceedings of the National Academy of Sciences, 2019, 116(1): 148-157. doi: 10.1073/pnas.1815345116 [26] TRIPATHI S, LEVINE H, JOLLY M K. The physics of cellular decision making during epithelial-mesenchymal transition[J]. Annual Review of Biophysics, 2020, 49(1): 1-18. doi: 10.1146/annurev-biophys-121219-081557 [27] PEINADO H, QUINTANILLA M, CANO A. Transforming growth factor β-1 induces snail transcription factor in epithelial cell lines: mechanisms for epithelial mesenchymal transitions[J]. Journal of Biological Chemistry, 2003, 278(23): 21113-21123. doi: 10.1074/jbc.M211304200 [28] DAVE N, GUAITA-ESTERUELAS S, GUTARRA S, et al. Functional cooperation between SNAIL1 and TWIST in the regulation of ZEB1 expression during epithelial to mesenchymal transition[J]. Journal of Biological Chemistry, 2011, 286(14): 12024-12032. doi: 10.1074/jbc.M110.168625 [29] CANO A, PÉREZ-MORENO M A, RODRIGO I, et al. The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression[J]. Nature Cell Biology, 2000, 2(2): 76-83. doi: 10.1038/35000025 [30] BATLLE E, SANCHO E, FRANCÍ C, et al. The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells[J]. Nature Cell Biology, 2000, 2(2): 84-89. doi: 10.1038/35000034 [31] MORENO-BUENO G, CUBILLO E, SARRIÓ D, et al. Genetic profiling of epithelial cells expressing E-cadherin repressors reveals a distinct role for snail, slug, and E47 factors in epithelial-mesenchymal transition[J]. Cancer Research, 2006, 66(19): 9543-9556. doi: 10.1158/0008-5472.CAN-06-0479 [32] VANDEWALLE C, COMIJN J, DE CRAENE B, et al. SIP1/ZEB2 induces EMT by repressing genes of different epithelial cell-cell junctions[J]. Nucleic Acids Research, 2005, 33(20): 6566-6578. doi: 10.1093/nar/gki965 [33] JIN G, ZHANG Z, WAN J, et al. G3BP2: structure and function[J]. Pharmacological Research, 2022, 186: 106548. doi: 10.1016/j.phrs.2022.106548 [34] FATTET L, JUNG H Y, MATSUMOTO M W, et al. Matrix rigidity controls epithelial-mesenchymal plasticity and tumor metastasis via a mechanoresponsive EPHA2/LYN complex[J]. Developmental Cell, 2020, 54(3): 302-316. doi: 10.1016/j.devcel.2020.05.031 [35] 龚博, 林骥, 王彦中, 等. 细胞骨架与细胞外基质的力学建模与分析[J]. 应用数学和力学, 2021, 42(10): 1024-1044. doi: 10.21656/1000-0887.420302GONG Bo, LIN Ji, WANG Yanzhong, et al. Mechanical modeling and analyses of cytoskeleton and extracellular matrix[J]. Applied Mathematics and Mechanics, 2021, 42(10): 1024-1044. (in Chinese) doi: 10.21656/1000-0887.420302 [36] CHENG B, WAN W, HUANG G, et al. Nanoscale integrin cluster dynamics controls cellular mechanosensing via FAKY397 phosphorylation[J]. Science Advances, 2020, 6(10): eaax1909. doi: 10.1126/sciadv.aax1909 [37] 程波, 徐峰. 考虑细胞外基质黏弹性行为的细胞黏附力学模型[J]. 应用数学和力学, 2021, 42(10): 1074-1080. doi: 10.21656/1000-0887.420259CHENG Bo, XU Feng. A molecular clutch model of cellular adhesion on viscoelastic substrate[J]. Applied Mathematics and Mechanics, 2021, 42(10): 1074-1080. (in Chinese) doi: 10.21656/1000-0887.420259 [38] ALEXANDER N R, TRAN N L, REKAPALLY H, et al. N-cadherin gene expression in prostate carcinoma is modulated by integrin-dependent nuclear translocation of TWIST1[J]. Cancer Research, 2006, 66(7): 3365-3369. doi: 10.1158/0008-5472.CAN-05-3401 [39] REN J, CROWLEY S D. TWIST1: a double-edged sword in kidney diseases[J]. Kidney Diseases, 2020, 6(4): 247-257. doi: 10.1159/000505188 [40] YANG M H, HSU D S, WANG H W, et al. BMI1 is essential in TWIST1-induced epithelial-mesenchymal transition[J]. Nature Cell Biology, 2010, 12(10): 982-992. doi: 10.1038/ncb2099 [41] XU Y, LEE D K, FENG Z, et al. Breast tumor cell-specific knockout of TWIST1 inhibits cancer cell plasticity, dissemination, and lung metastasis in mice[J]. Proceedings of the National Academy of Sciences, 2017, 114(43): 11494-11499. doi: 10.1073/pnas.1618091114 [42] CASAS E, KIM J, BENDESKY A, et al. SNAIL2 is an essential mediator of TWIST1-induced epithelial mesenchymal transition and metastasis[J]. Cancer Research, 2011, 71(1): 245-254. doi: 10.1158/0008-5472.CAN-10-2330 [43] TRAN D D, CORSA CAS, BISWAS H, et al. Temporal and spatial cooperation of SNAIL1 and TWIST1 during epithelial-mesenchymal transition predicts for human breast cancer recurrence[J]. Molecular Cancer Research, 2011, 9(12): 1644-1657. doi: 10.1158/1541-7786.MCR-11-0371 [44] SUBBALAKSHMI A R, ASHRAF B, JOLLY M K. Biophysical and biochemical attributes of hybrid epithelial/mesenchymal phenotypes[J]. Physical Biology, 2022, 19(2): 025001. doi: 10.1088/1478-3975/ac482c [45] CUI J, ZHANG C, LEE J E, et al. MLL3 loss drives metastasis by promoting a hybrid epithelial-mesenchymal transition state[J]. Nature Cell Biology, 2023, 25(1): 145-158. doi: 10.1038/s41556-022-01045-0 [46] MULLINS R D Z, PAL A, BARRETT T F, et al. Epithelial-mesenchymal plasticity in tumor immune evasion[J]. Cancer Research, 2022, 82(13): 2329-2343. doi: 10.1158/0008-5472.CAN-21-4370 [47] ZHU H, MIAO R, WANG J, et al. Advances in modeling cellular mechanical perceptions and responses via the membrane-cytoskeleton-nucleus machinery[J]. Mechanobiology in Medicine, 2024, 2(1): 100040. doi: 10.1016/j.mbm.2024.100040 [48] ZHANG C, ZHU H, REN X, et al. Mechanics-driven nuclear localization of YAP can be reversed by N-cadherin ligation in mesenchymal stem cells[J]. Nature Communications, 2021, 12(1): 6229. doi: 10.1038/s41467-021-26454-x [49] ZHANG Z, ZHU H, ZHAO G, et al. Programmable and reversible integrin-mediated cell adhesion reveals hysteresis in actin kinetics that alters subsequent mechanotransduction[J]. Advanced Science, 2023, 10(35): 2302421. doi: 10.1002/advs.202302421 -




 下载:
下载:









 渝公网安备50010802005915号
渝公网安备50010802005915号